![]()
Register and administer comprehensive risk assessment
![]()
It allows investigators to capture data on vaccine administered and doses
![]()
The platform allows periodic follow-up post-vaccination for vaccine adverse events (AESI)
![]()
Interactive charts allows visualisation of data in interactive chart and graphs
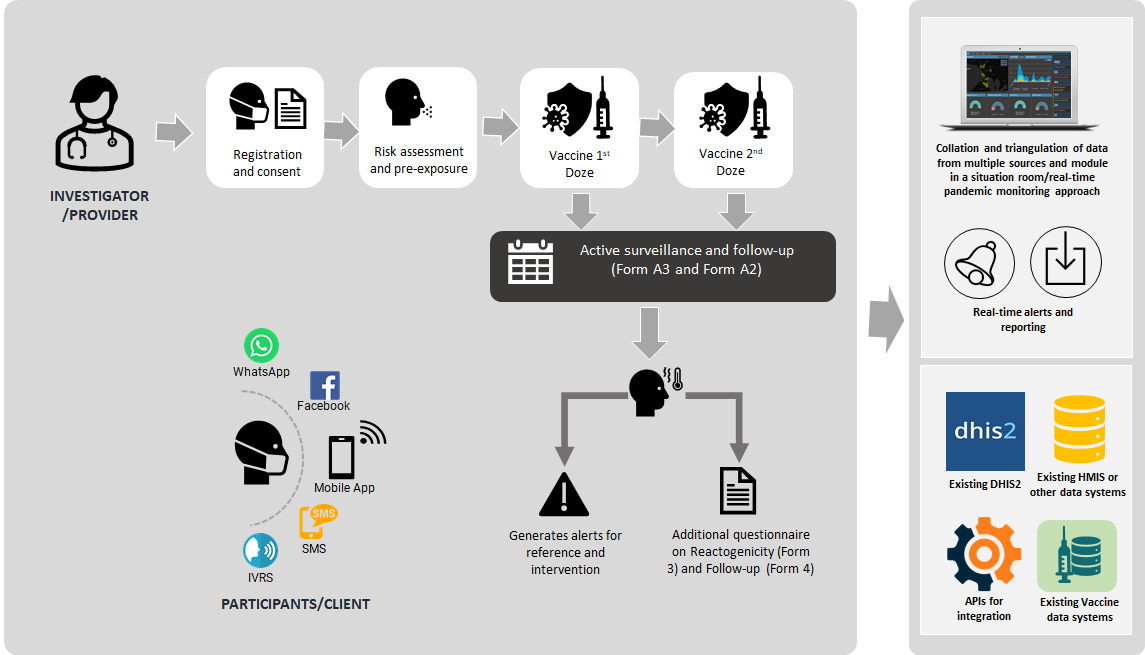 Fig 1: Workflow diagram demonstrating a comprehensive ready-to-use digital package for countries to implement and harmonize their vaccine safety and adverse event reporting initiatives
Fig 1: Workflow diagram demonstrating a comprehensive ready-to-use digital package for countries to implement and harmonize their vaccine safety and adverse event reporting initiatives
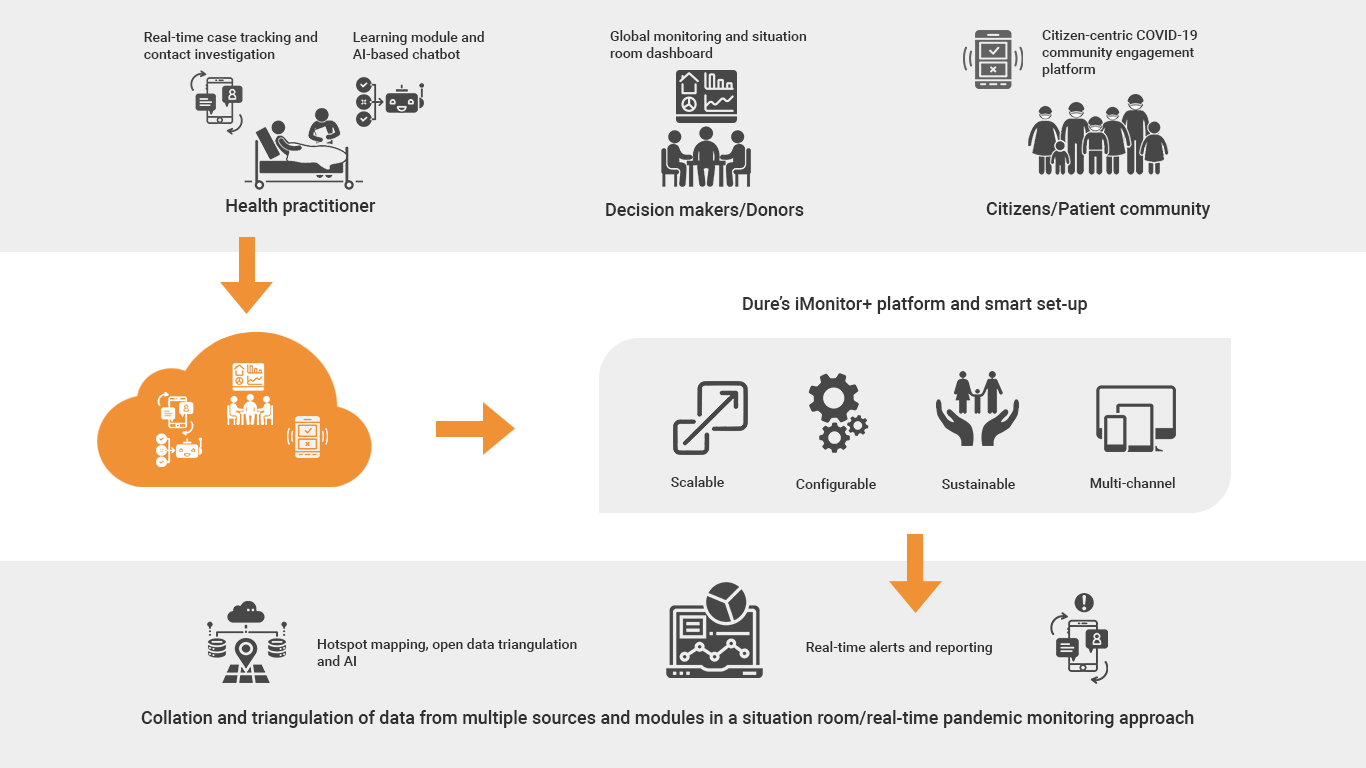
Dure Technologies (Dure) is a global leader in providing digital solutions in public health. In association with WHO, Dure has been assisting multiple countries in their Vaccine Safety initiatives. Recently Dure supported India in one of the largest COVID-19 Vaccine Safety initiatives, which is currently being rolled out by more than 1 million facilities across the country.
Dure has its footprints in more than 40 countries and has implemented more than 100+ digital innovation projects for UN agencies like WHO, UNAIDS, UNICEF, UNOPS, Ministries of Health and large donors in the area of immunization, COVID, HIV, TB and many other public domain. Dure has an established network of on-site resources and local country partnerships to support these digital initiatives on the ground.
Dure Technologies is a pioneer Organization, which focuses on providing innovative Digital Health solutions for social impact.
Dure has also been associated with WHO at a strategic level which also includes Long Term Agreement (LTA) from 2020-2024 with WHO for the provisioning of strategic leadership assistance, digital health technology and innovation assessment, incubation and scale up, development of strategies, policies and investment plans. Dure Technologies is a pioneer Organization, which focused on providing innovative Digital Health solutions for social impact.
Apart from having a strong technical expertise, Dure follows a strong implementation science and design thinking approach to ensure successful implementation of these digital platforms in the countries.
![]()
We will help schedule a live demonstration of the platform and discuss country adaptation and harmonisation needs
![]()
A simple step by step onboarding process finalisation of Technical Assistance model
![]()
Comprehensive digital platform and implementation package made available to the countries
Rue de Lausanne 37, 1201 Geneva, Switzerland
1101/1102, Centrum IT Park, Opp. TMC Office Wagle Estate,Thane west- 400602
"EMPOWERERS", 2nd & 3rd Floor, Saira Tower, Gulmohar Commercial Complex, New Delhi -110049
63 Bartholomew Street, Newbury, England, RG14 7BE